 
The downside if there is one is that this method does need a little bit of intuition. It’s the best method I know of for balancing extremely difficult equations and is a very fast method for balancing hard redox equations too, once you get the hang of it. This method does exactly that – it uses logic to reduce the number of unknowns you need to solve. Now, if in the above example you were thinking “ hang on, if a = c = d, why introduce c and d at all? Why not simplify the whole thing?” you would have been right and this is where the simplified algebraic method comes in. Simple algebraic method for balancing chemical equations This is perfectly fine, there is no right or wrong approach. Note that when you use the algebraic method, you may end up performing different substitutions in order to eliminate the unknowns. In the unlikely event this showed up on an exam paper as a balancing question, you would ordinarily use the redox balancing method but I think you’d agree that this method is faster. 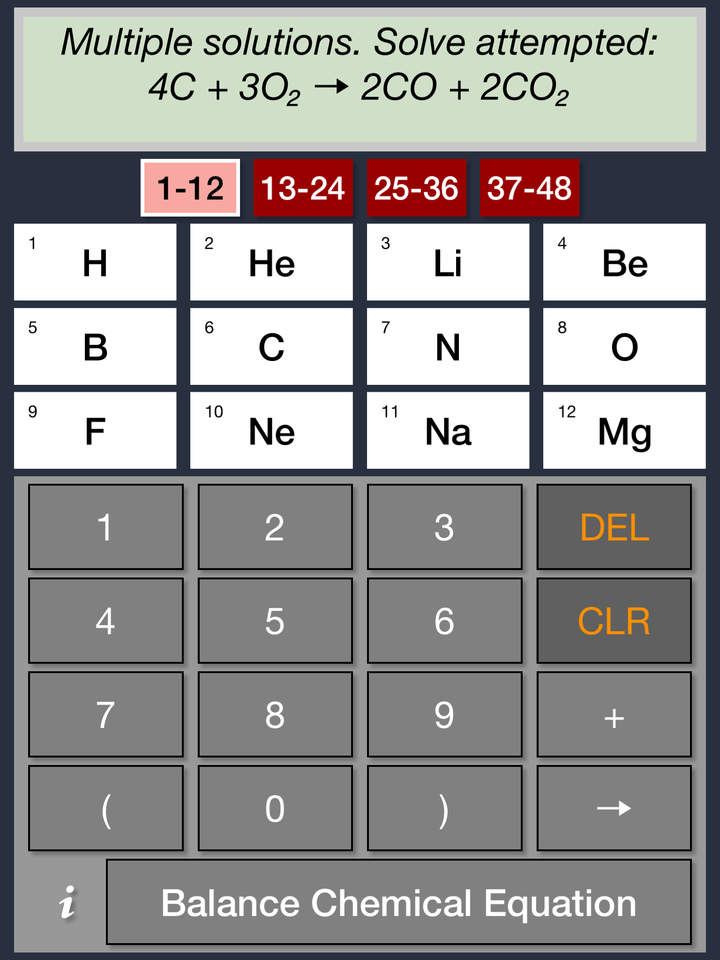
Having now found two coefficients, substituting into the rules for Cl and O and using the fact that a = c = d = 2 solves for b and f:ĢKMnO 4 + 16HCl → 2MnCl 2 + 2KCl + 5Cl 2 + 8H 2O We can also get rid of b using the rule for H:Īnd finally, get rid of f using the rule for O: There are too many unknowns here, but we can substitute the rules for K and Mn into the rule for Cl to get rid of c and d: On the product side, MnCl 2 contains two chlorine atoms, so if its coefficient is c, it must contain 2c chlorine atoms, whilst KCl contains d chlorine atoms and so on, adding up the total number of chlorine atoms on the RHS. On the reactant side, we would have a total of b chlorine atoms. To explain the logic behind this using Cl as an example, we know that the number of chlorine atoms must be the same on both sides of the equation. Next, applying the Conservation of Mass, which tell us that the total number of atoms of each element must be the same on both sides, write algebraic rules for each element. _KMnO 4 + _HCl → _MnCl 2 + _KCl + _Cl 2 + _H 2Oįirst thing we do is give each compound a letter coefficient:ĪKMnO 4 + bHCl → cMnCl 2 + dKCl + eCl 2 + fH 2O 
That makes absolutely no sense I’m sure without an example, so here’s an equation to balance using this strategy: Substitute the values into the other rules to obtain the balancing coefficients.Substitute and simplify to obtain a rule that equates only two letter coefficients that you can solve.Write algebraic expressions or rules for each element that equate its atoms on the LHS and RHS.Write a different letter coefficient in front of each compound in the equation.The strategy for balancing chemical equations algebraically is as follows: Post memes/jokes in /r/chemistrymemes and /r/chemistryjokes.Algebraic method for balancing chemical equations Any such posts will be deleted.Īsk education and jobs questions in the current weekly topic. If you're looking for a more concentrated, advanced discussion of chemistry topics among professionals and grad students, check out /r/Chempros.īefore asking "What chemical is this?" see this chart. Click here for the OSHA chemical data site and here for a multicompany MSDS aggregate search. If you spill/injure yourself contact medical professionals and read the MSDS, do not post to this reddit. Yes links to blogs, images, videos, comics, and infographics are okay especially if they are on your personal website. No physorg, sciencedaily, or other press release aggregator spam! If a caption or explanation is included this helps, but please use your discretion.īefore asking about chemical drawing/illustration programs, look at your school's IT/software website and see if they provide an institutional license of ChemDraw (hint: if they have a chemistry department, they will) Likewise, simple pictures of uninteresting and garden variety chemistry-related things are not appreciated. No memes, rage comics, image macros, reaction gifs, or other "zero-content" material. However, academic discussions on pharmaceutical chemistry and the science of explosives are permitted. Rules: Violating a rule will result in a ban.Īsk homework, exam, lab, and other undergraduate-level questions at ChemicalForums otherwise it will be deleted.ĭiscussions on illicit drug synthesis, bomb making, and other illegal activities are not allowed and will lead to a ban. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
